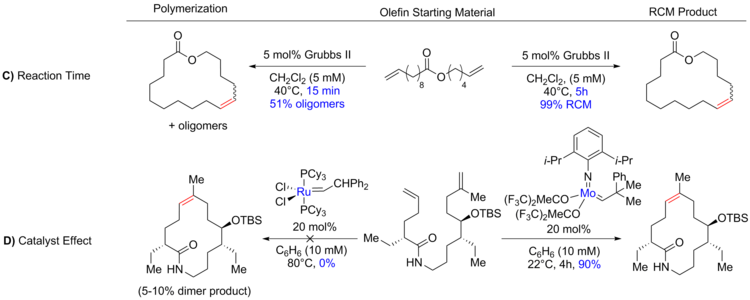
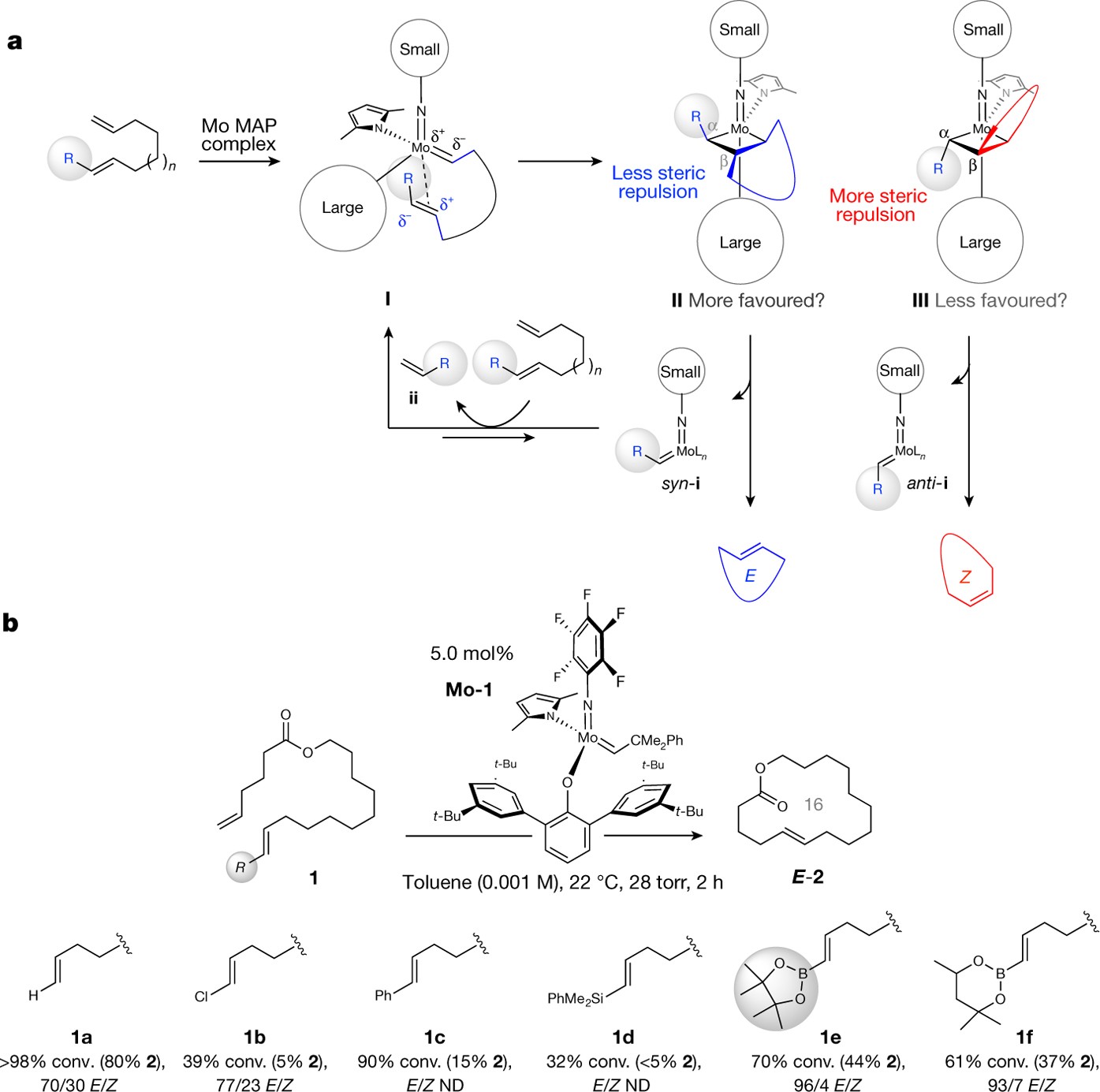
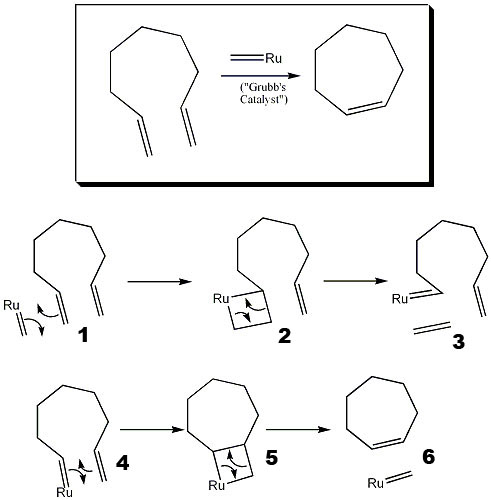
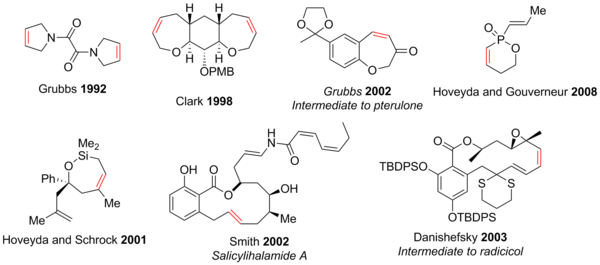
Tandem Ring‐Opening–Ring‐Closing Metathesis for Functional Metathesis Catalysts - Nagarkar - 2016 - Angewandte Chemie International Edition - Wiley Online Library

Ring Closing Alkyne Metathesis. Comparative Investigation of Two Different Catalyst Systems and Application to the Stereoselective Synthesis of Olfactory Lactones, Azamacrolides, and the Macrocyclic Perimeter of the Marine Alkaloid Nakadomarin A

Ring-Closing Metathesis of Allylsilanes/Electrophilic Desilylation To Prepare exo-Methylidenecycloalkanes. Short Syntheses of Teucladiol and Poitediol | Journal of the American Chemical Society
![Selective ring-rearrangement or ring-closing metathesis of bicyclo[3.2.1]octenes - Chemical Communications (RSC Publishing) DOI:10.1039/D0CC04624H Selective ring-rearrangement or ring-closing metathesis of bicyclo[3.2.1]octenes - Chemical Communications (RSC Publishing) DOI:10.1039/D0CC04624H](https://pubs.rsc.org/image/article/2020/CC/d0cc04624h/d0cc04624h-s2_hi-res.gif)
Selective ring-rearrangement or ring-closing metathesis of bicyclo[3.2.1]octenes - Chemical Communications (RSC Publishing) DOI:10.1039/D0CC04624H